Specialising in custom-designed, precision scientific instruments, built, programmed and calibrated
to the most exacting standards. The range includes precision dataloging barographs,
with built-in statistical analysis, Barographic Transient Event Recorders
and computer-interfaced detectors and sensors
for environmental monitoring & process control.

A site dedicated to scientific techniques, experimental methods, &
investigative tools for the inventor, researcher
and laboratory pioneer. Articles on glassblowing, electronics, metalcasting, magnetic
measurements with new material added continually. Check it out!
www.drkfs.net
Sample Solvents
Ideally, solvents used for dissolving a sample for
NMR examination should not contain the element that is being
examined. Consequently, if proton NMR was being employed then carbon
tetrachloride or carbon disulphide would be appropriate solvents.
Unfortunately, these are exclusively dispersive
solvents in which polar solutes are relatively insoluble. An
alternative a solvent that had only one single proton absorption
might be useful and such solvents would include chloroform, acetone,
benzene, dimethylsuphoxide , water, dioxane methylenechloride and
trifluoroacetic acid etc. Even better, the deuterated isomers of the
above solvents could be used but deuterated solvents can be somewhat
expensive.
Solvents can affect the NMR Spectrum produced as the
proton shifts can often be affected by the nature of the surrounding
solvent molecules. The interacting molecules can also be, in fact,
those of the surrounding molecules of the pure sample itself. This
interference can arise from two sources; the first is the effect of
changes in diamagnetic susceptibility of the solute or solvent and
the other is the effect of solute/solute and solute/solvent
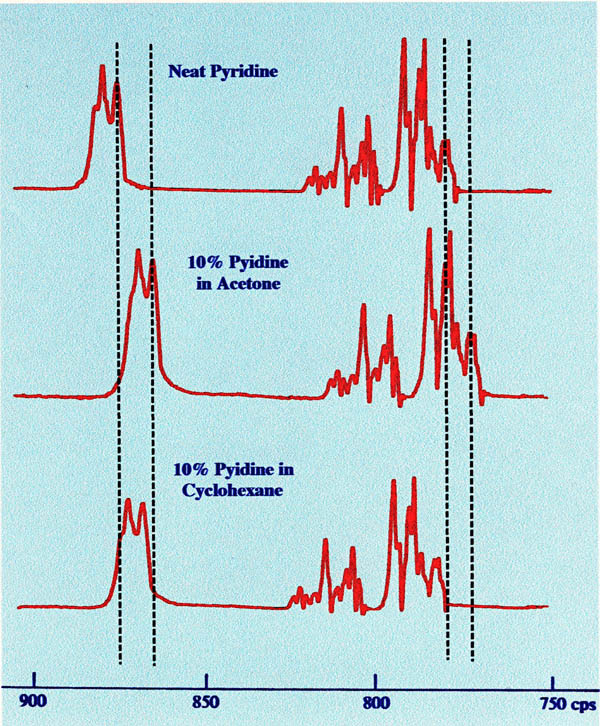
interactions. Figure 10 shows the change in chemical shift of the
pyridine protons when examined as a pure solvent and as 10% solutions
of pyridine in acetone and cyclohexane .
Chemical shifts can also be affected
by concentration changes and in addition temperature changes. These
changes are generally small except for protons attached to oxygen ,
sulphur and nitrogen atoms. For example, a temperature change from
–118oC
to +75oC can
cause the hydroxyl protons of ethanol to shift 1.5 ppm. The effect of
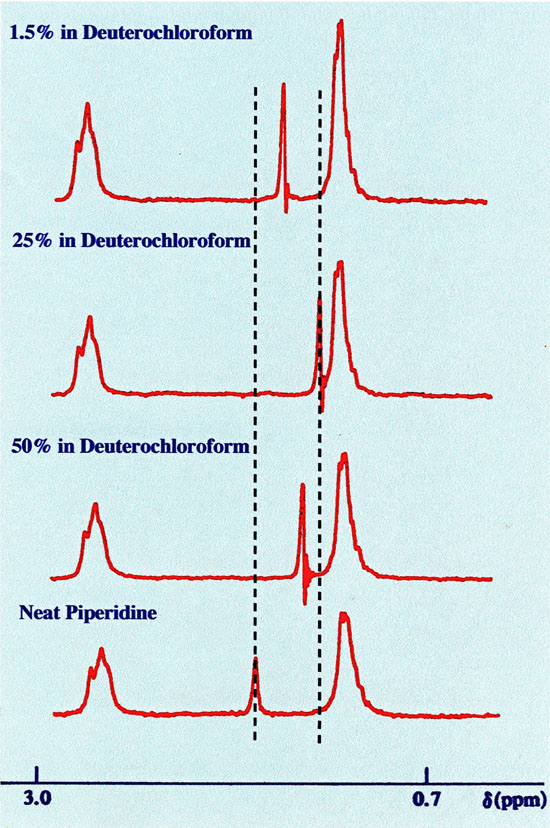
sample concentration on chemical shift is demonstrated clearly in
figure 11.
The greatest shift change occurs with the singlet, which becomes
more shielded with the presence of the solvent and its increase from
25 to 50% of the sample. However, there appears to be some anomaly as
the N-H proton appears to be more shielded in the 1.5% solution.
Another interesting solution effect
is the proton replacement in such groups as OH, NH, SH and similar
groups. The protons in these groups can easily be replaced by
deuterium
e.g.
R-XH +D2O = R-XD +DHO
It is clear that the deuterium oxide
must be present in adequate quantity so that the equilibrium is in
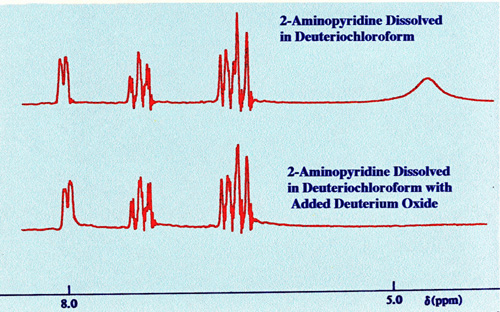
favour of the proton replacement. An example is given in figure 12
where the proton in the N-H group of 2-aminopyridine is replaced by
deuterium employing deuterium oxide in the sample solution.
The proton shifts of many aromatic
compounds are strongly affected by concentration. Extrapolating the
chemical shift of benzene in cyclohexane from 80% to infinite
dilution will result in a shift of –0.3 ppm. The converse also
applies for cyclohexane dissolved in benzene . The cause is the large
diamagnetic anisotropy of aromatic molecules caused by the
circulation of the ‘π’
electrons forming a ‘ring current’. This causes protons
proximate to the ring centre to be shielded. These problems are not
frequent but they should be kept in mind in the practice of NMR
spectroscopy. From practical experience, the following general
conclusions have been made with regard to benzene as a solute/solvent
A benzene
nucleus will preferentially interact directly at electron deficient
sites in any solvent molecule
Solute
interaction with the benzene molecule can occur as a 1-1 association
and transiently at any electron deficient site.
The
orientation of the benzene molecule is a result of induced
dipole polarization
As a result of polarization the positive charge
resulting from the induced dipole will normally be situated far from
the negative end of the dipole.

About the Author
RAYMOND PETER WILLIAM SCOTT was born on June 20 1924 in Erith, Kent, UK. He studied at the
University of London, obtaining his B.Sc. degree in 1946 and his D.Sc. degree in 1960.
After spending more than a decade at Benzole Producers, Ltd. Where he became head of
the Physical Chemistry Laboratory, he moved to Unilever Research Laboratories as
Manager of their Physical Chemistry department. In 1969 he became Director of Physical
Chemistry at Hoffmann-La Roche, Nutley, NJ, U.S.A. and subsequently accepted the position
of Director of the Applied Research Department at the Perkin-Elmer Corporation, Norwalk, CT, U.S.A.
In 1986 he became an independent consultant and was appointed Visiting Professor at Georgetown
University, Washington, DC, U.S.A. and at Berkbeck College of the University of London; in 1986
he retired but continues to write technical books dealing with various aspects of physical chemistry
and physical chemical techniques. Dr. Scott has authored or co-authored over 200 peer reviewed
scientific papers and authored, co-authored or edited over thirty books on various aspects of
physical and analytical chemistry. Dr. Scott was a founding member of the British chromatography
Society and received the American Chemical society Award in chromatography (1977), the
M. S. Tswett chromatography Medal (1978), the Tswett chromatography Medal U.S.S.R., (1979),
the A. J. P. Martin chromatography Award (1982) and the Royal Society of Chemistry Award in
Analysis and Instrumentation (1988).
Dr. Scott’s activities in gas chromatography started at the inception of the technique,
inventing the Heat of Combustion Detector (the precursor of the Flame Ionization Detector),
pioneered work on high sensitivity detectors, high efficiency columns and presented fundamental
treatments of the relationship between the theory and practice of the technique.
He established the viability of the moving bed continuous preparative gas chromatography,
examined both theoretically and experimentally those factors that controlled dispersion
in packed beds and helped establish the gas chromatograph as a process monitoring instrument.
Dr. Scott took and active part in the renaissance of liquid chromatography,
was involved in the development of high performance liquid chromatography and invented
the wire transport detector. He invented the liquid chromatography mass spectrometry
transport interface, introduced micro-bore liquid chromatography columns and used them
to provide columns of 750,000 theoretical plates and liquid chromatography separations
in less than a second.
Dr. Scott has always been a “hands-on” scientist with a remarkable record of accomplishments in chromatography ranging from hardware design to the development of fundamental theory. He has never shied away from questioning “conventional wisdom” and his original approach to problems has often produced significant breakthroughs.





